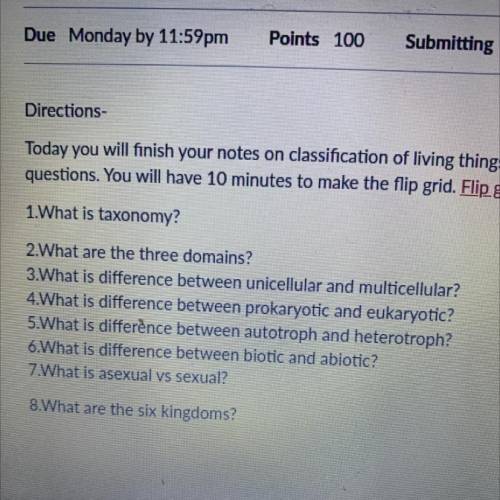
I need help pretty bad can you answer all these questions
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 02:00
What causes the appearance of lines in a emission spectrum
Answers: 1

Chemistry, 23.06.2019 06:00
Is the flow of energy during vaporizing more like the flow during melting or during freezing
Answers: 1

Chemistry, 23.06.2019 21:20
Yasmin's teacher asks her to make a supersaturated saline solution. her teacher tells her that the solubility of the salt is 360 g/l at room temperature (25 °c). how can yasmin make a supersaturated saline solution? she can add 380 g of salt to 1 l of hot water (75 °c) and stir until all the salt dissolves. then, she can carefully cool the solution to room temperature (25 °c). she can add 360 g of salt to 1 l of room temperature water (25 °c) and stir the solution until all the salt dissolves. she can add 380 g of salt to 1 l of cold water (5 °c) and stir the solution until most of the salt dissolves. then, she can carefully heat the solution to room temperature (25 °c). she can add 380 g of salt to 1 l of room temperature water (25 °c) and let the solution sit for 24 hours, so the salt dissolves.
Answers: 2
You know the right answer?
Questions



Social Studies, 26.08.2019 04:00

Social Studies, 26.08.2019 04:00

Biology, 26.08.2019 04:00

Spanish, 26.08.2019 04:00

Mathematics, 26.08.2019 04:00

Mathematics, 26.08.2019 04:00

Spanish, 26.08.2019 04:00





History, 26.08.2019 04:00

Mathematics, 26.08.2019 04:00




English, 26.08.2019 04:00