
3.04 Quiz: Chemical Reactions 1 to 10 question
question 1 to 8 is on first page Graphic Organizer: Use the terms in the word bank to complete the graphic organizer below.
question 9 to 10 on second page
Is this a chemical reaction or not? Explain why you think so (Be sure to answer both parts of the question
9.
Summarize your understanding: using everything you have just reviewed above, explain what a chemical reaction is and how a person would know whether a chemical reaction has occurred or not.
ill give brainliest
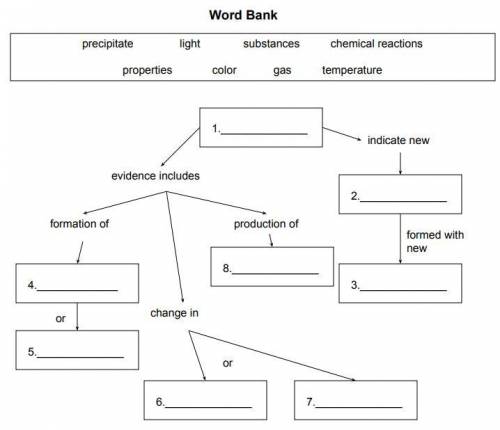
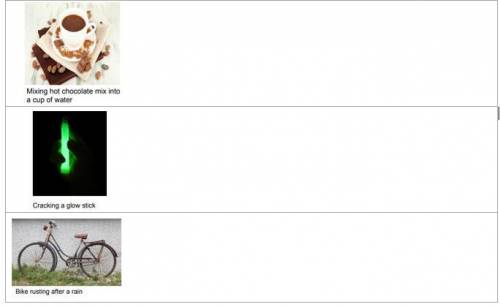

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Problem #3 (ch. 1, problem 15)the ideal gas law provides one way to estimate the pressure exerted by a gas on a container. the law isí‘ťí‘ť=푛푛푛푛푛푛푉푉more accurate estimates can be made with the van der waals equationí‘ťí‘ť=푛푛푛푛푛푛푉푉â’푛푛푟푟â’푞푞푛푛2푉푉2where the term nb is a correction for the volume of the molecules and the term an2/v2is a correction for molecular attractions. the values of a and b depend on the type of gas. the gas constant is r, the absolutetemperature is t, the gas volume is v, and the number of moles of gas molecules is indicated by n. if n = 1 mol of an ideal gas were confined to a volume of v = 22.41 l at a temperature of 0â°c (273.2k), it would exert a pressure of 1 atm. in these units, r = 0.0826.for chlorine gas (cl2), a = 6.49 and b = 0.0562. compare the pressure estimates given by the ideal gas law and the van der waals equation for 1 mol of cl2 in 22.41 l at 273.2 k. what is the main cause of the difference in the two pressure estimates, the molecular volume or the molecular attractions?
Answers: 1

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1


Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1
You know the right answer?
3.04 Quiz: Chemical Reactions 1 to 10 question
question 1 to 8 is on first page Graphic Organizer:...
Questions

Physics, 30.06.2019 10:30



History, 30.06.2019 10:30


Mathematics, 30.06.2019 10:30



Physics, 30.06.2019 10:30


Mathematics, 30.06.2019 10:30





Mathematics, 30.06.2019 10:30

Arts, 30.06.2019 10:30

Mathematics, 30.06.2019 10:30




