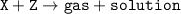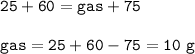Chemistry, 12.11.2020 04:40 tayleeanntabeln2226
A gas is produced when 25 g of substance X is mixed into 60 g of solution Z in a beaker. The mass of the solution in the beaker after the reaction is 75 g. What is the mass of the gas released? (X + Z -> gas + solution)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2

Chemistry, 22.06.2019 22:10
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1

Chemistry, 23.06.2019 09:30
What is the best describtion of the side of the moon that faces earth?
Answers: 2

Chemistry, 23.06.2019 10:30
Which of the following characteristics are true of enzymes? check all that apply. a.)the structure of an enzyme can change if conditions change. b.)a single enzyme can normally catalyze a wide variety of reactions under many conditions. c.)enzymes are found only in nonliving systems. d.)enzymes allow living things to regulate body conditions through feedback mechanisms. e.)enzymes bind to specific substrates in specific ways. f.)enzymes increase the rate of reaction. g.)when shown in energy-reaction diagrams, enzymes represent the higher activation energy.
Answers: 1
You know the right answer?
A gas is produced when 25 g of substance X is mixed into 60 g of solution Z in a beaker. The mass of...
Questions


Mathematics, 05.03.2021 18:50

English, 05.03.2021 18:50



Mathematics, 05.03.2021 18:50


Mathematics, 05.03.2021 18:50

Mathematics, 05.03.2021 18:50


History, 05.03.2021 18:50


Mathematics, 05.03.2021 18:50


Mathematics, 05.03.2021 18:50


Social Studies, 05.03.2021 18:50