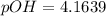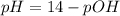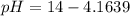Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 21:30
What is the effect of returning nuclear reactor cooling water back into bodies of water?
Answers: 3

Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3

Chemistry, 23.06.2019 05:30
Elizabeth has two separate samples of the same substance. sample is in the liquid state, and the other is in the solid state. the two samples most likely differ in which property?
Answers: 1
You know the right answer?
Calculate the pH during the titration of 20.00 mL of 0.1000 M trimethylamine, (CH3)3N(aq), with 0.20...
Questions




History, 18.11.2020 07:50

Computers and Technology, 18.11.2020 07:50



History, 18.11.2020 07:50


Mathematics, 18.11.2020 07:50

Social Studies, 18.11.2020 07:50

Biology, 18.11.2020 07:50


English, 18.11.2020 07:50

Geography, 18.11.2020 07:50


Mathematics, 18.11.2020 07:50

History, 18.11.2020 07:50

History, 18.11.2020 07:50

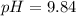
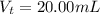
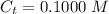
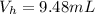
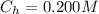
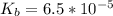
![pOH = pK_b + log [\frac{N_h}{N_b} ]](/tpl/images/0887/8286/4d586.png)
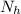 is the number of moles of acid which is evaluated as
is the number of moles of acid which is evaluated as 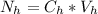
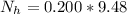
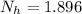
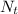 is the number of moles of acid which is evaluated as
is the number of moles of acid which is evaluated as 
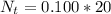
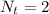
![pOH = -log(K_b) + log [\frac{N_h}{N_b} ]](/tpl/images/0887/8286/f69f7.png)
![pOH = -log(6.5*10^{-5}) + log [\frac{1.896}{2} ]](/tpl/images/0887/8286/49d4f.png)