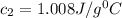Chemistry, 06.11.2020 16:30 halltristan657
You are performing an experiment where you place 6.7797 g of Al metal at 205.24 equationC into a coffee cup calorimeter that contains 87.4 g of water at 25.37 equationC. The final temperature of the water in the coffee cup is 28.67 equationC. Remember, the heat gained by the water is equal to the heat lost by the Al. equation What is the specific heat (C) of the Al in J/g°C? Cwater= 4.184 J/g°C. Do not include units. If you need to express your answer as an exponential number, use this template: 1445 should be typed as 1.445e+003

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 22.06.2019 03:00
Flourine is found to undergo 10% radioactivity decay in 366 minutes determine its halflife
Answers: 3

Chemistry, 22.06.2019 09:40
In the lab, ammonia was mixed with water to form ammonium hydroxide. what is/are the reactant(s)? o water and ammonia o ammonia o ammonium hydroxide need
Answers: 2

Chemistry, 22.06.2019 19:00
Avolleyball player hit a ball with a mass of 0.25 kg. the average acceleration of the ball is 15.5 m/s². how much force did the volleyball player apply to the ball? 62.0 n 3.87 n 62.0 m/s² 3.87 m/s²
Answers: 2
You know the right answer?
You are performing an experiment where you place 6.7797 g of Al metal at 205.24 equationC into a cof...
Questions

Computers and Technology, 03.12.2020 20:10




Chemistry, 03.12.2020 20:10



Spanish, 03.12.2020 20:10


Chemistry, 03.12.2020 20:10


Mathematics, 03.12.2020 20:10







Mathematics, 03.12.2020 20:10

Mathematics, 03.12.2020 20:10

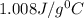 .
.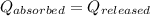
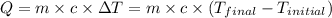
![m_1\times c\times (T_{final}-T_1)=-[m_2\times c\times (T_{final}-T_2)]](/tpl/images/0874/2122/b0e58.png)
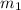 = mass of water = 87.4 g
= mass of water = 87.4 g 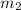 = mass of Al metal = 6.7797 g
= mass of Al metal = 6.7797 g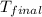 = final temperature =
= final temperature = 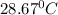
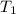 = temperature of water =
= temperature of water = 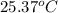
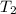 = temperature of Al metal =
= temperature of Al metal = 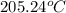
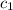 = specific heat of water =
= specific heat of water = 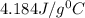
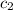 = specific heat of Al metal = ?
= specific heat of Al metal = ? ![m_1\times c_1\times (T_{final}-T_1)=-[m_2\times c_2\times (T_{final}-T_2)]](/tpl/images/0874/2122/09236.png)
![87.4\times 4.184\times (28.67-25.37)^0C=-[6.7797\times c_2\times (28.67-205.24)]](/tpl/images/0874/2122/2d990.png)