25 pts
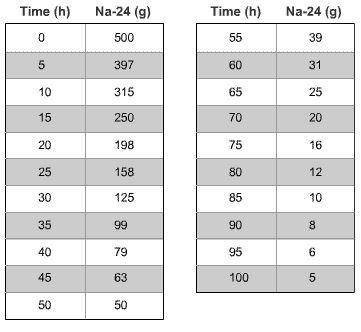
2. The data in the table show the amount of a 500 g sample of sodium-24 over time.
Ma...

Chemistry, 05.11.2020 22:00 daniel7363
25 pts
2. The data in the table show the amount of a 500 g sample of sodium-24 over time.
Make a graph of the data (remember to label all axes and title the graph).
What is the half-life of sodium-24? Explain how you determined your answer.
Sodium-24 decays by beta emission. Write a nuclear equation that shows the decay of sodium-24 and
its products

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 22.06.2019 21:20
One way in which the useful metal copper is produced is by dissolving the mineral azurite, which contains copper(ii) carbonate, in concentrated sulfuric acid. the sulfuric acid reacts with the copper(ii) carbonate to produce a blue solution of copper(ii) sulfate. scrap iron is then added to this solution, and pure copper metal precipitates out because of the following chemical reaction: (s) (aq) (s) (aq) suppose an industrial quality-control chemist analyzes a sample from a copper processing plant in the following way. he adds powdered iron to a copper(ii) sulfate sample from the plant until no more copper will precipitate. he then washes, dries, and weighs the precipitate, and finds that it has a mass of .
Answers: 2

Chemistry, 23.06.2019 06:30
What type of chemical reaction occurs between silver nitrate (agno3) and copper (cu)? the equation i was given is 2agno3 + cu —> 2ag+ cu(no3)2.
Answers: 1
You know the right answer?
Questions


Biology, 03.12.2021 21:40




Social Studies, 03.12.2021 21:40


Arts, 03.12.2021 21:40

Biology, 03.12.2021 21:40



Computers and Technology, 03.12.2021 21:40

Mathematics, 03.12.2021 21:40

English, 03.12.2021 21:40



English, 03.12.2021 21:40





