
Chemistry, 05.11.2020 18:50 andreanaapollon7593
A compound containing Na, C, and O is found to have 1.06 mol Na, 0.528 mol C, and 1.59 mol O. What is the empirical formula of the compound?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 10:00
Main expenses you plan on making payments on a new car too. you want to spend 15% of your monthly net pay on the car payment, insurance, registration, and taxes combined. what is your monthly car allowance? $149.46 $298.91 $448.37 $597.83
Answers: 3

Chemistry, 22.06.2019 16:00
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1
You know the right answer?
A compound containing Na, C, and O is found to have 1.06 mol Na, 0.528 mol C, and 1.59 mol O. What i...
Questions


Mathematics, 26.03.2021 21:30



English, 26.03.2021 21:30

Physics, 26.03.2021 21:30




Mathematics, 26.03.2021 21:30



Mathematics, 26.03.2021 21:30

History, 26.03.2021 21:30

History, 26.03.2021 21:30

History, 26.03.2021 21:30




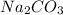 .
.

