
Chemistry, 04.11.2020 19:00 aredwolf2017
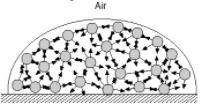
Surface tension is the inward pull that tends to minimize the surface area of a liquid. Water has high surface tension because of hydrogen bonding. In the diagram below, consider a molecule within the bulk of the liquid. This molecule experiences attractions to its neighboring molecules in all directions. These forces, shown by the arrows, average out to zero and there is no net force on the molecule. How is the situation different for a molecule at the surface? How does this give rise to surface tension?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 23.06.2019 02:40
How can a mixture of salt water be separated into salt and water
Answers: 1
You know the right answer?
Surface tension is the inward pull that tends to minimize the surface area of a liquid. Water has hi...
Questions


Geography, 07.10.2020 08:01



Chemistry, 07.10.2020 08:01


Advanced Placement (AP), 07.10.2020 08:01


Health, 07.10.2020 08:01





Mathematics, 07.10.2020 08:01

English, 07.10.2020 08:01

Biology, 07.10.2020 08:01






