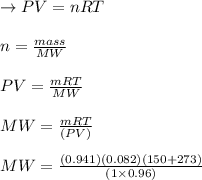A sample of an unknown compound is vaporized at . The gas produced has a volume of at a pressure of , and it weighs . Assuming the gas behaves as an ideal gas under these conditions, calculate the molar mass of the compound. Be sure your answer has the correct number of significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 00:00
#20 which type of bond is formed when bases pair in dna? ionic bond covalent bond coordinate bond hydrogen bond
Answers: 1

Chemistry, 23.06.2019 00:00
How do you determine the percent yield of a chemical reaction
Answers: 1

Chemistry, 23.06.2019 04:31
How many grams of iron can be made from 16.5 grams of fe2o3
Answers: 1

Chemistry, 23.06.2019 05:40
Convert a speed of 201 cm/s to units of inches per minute. also, show the unit analysis by dragging components into the unit‑factor slots.
Answers: 1
You know the right answer?
A sample of an unknown compound is vaporized at . The gas produced has a volume of at a pressure of...
Questions







Mathematics, 23.07.2019 23:30

History, 23.07.2019 23:30

Mathematics, 23.07.2019 23:30



Biology, 23.07.2019 23:30


History, 23.07.2019 23:30



Mathematics, 23.07.2019 23:30

History, 23.07.2019 23:30


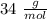 "
"