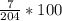Chemistry, 02.11.2020 17:00 landonp101
Calculate the percent mass per volume, % (m/v), of a dextrose solution containing 7.00 g of dextrose in 2.00×102 mL of solution. Note that mass is not technically the same as weight, but the abbreviation % (w/v) is often used interchangeably with % (m/v).

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 21:30
If you burn 46.6 g of hydrogen and produce 416 g of water, how much oxygen reacted
Answers: 3

Chemistry, 23.06.2019 01:00
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3

Chemistry, 23.06.2019 04:10
What does the field of thermodynamics relate to a-changes in nuclear reactions b- changes in energy in systems c changes in molecular structure d changes in atomic properties
Answers: 1
You know the right answer?
Calculate the percent mass per volume, % (m/v), of a dextrose solution containing 7.00 g of dextrose...
Questions

Advanced Placement (AP), 03.09.2020 23:01






History, 03.09.2020 23:01



Mathematics, 03.09.2020 23:01






History, 03.09.2020 23:01

Mathematics, 03.09.2020 23:01


Mathematics, 03.09.2020 23:01

English, 03.09.2020 23:01