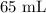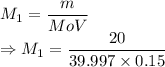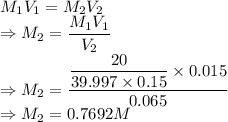Chemistry, 30.10.2020 16:50 kaelah6846
A student prepared a stock solution by dissolving 20.0 g of NaOH in enough water to make 150. mL of solution. She then took 15.0 mL of the stock solution and diluted it with enough water to make 65.0 mL of a final solution. What is the concentration of NaOH for the final solution?
A) O. 411 M
B) 0.534 M
C) 1.87 M
D) 2.43 M

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 02:30
Needthe meter is the standard unit for: 1) height 2) length 3) weight 4) mass
Answers: 3

Chemistry, 22.06.2019 02:30
Margaret wants to make an orange flavored drink by stirring powdered drink mix into a glass of water. she doesn't like drinks that have small clumps of powdered solid in them, so she wants the drink to be a perfect solution. what factors should margaret not consider when deciding how much powder to add to her glass of water?
Answers: 3

Chemistry, 22.06.2019 12:30
Nebulae are enormous clouds in outer space. they are made mostly of hydrogen gas, helium gas, and dust. some nebulae glow brightly, while others do not. the stars that people see are huge, bright balls of glowing gas. they are made mostly of hydrogen and helium. which statement correctly describes other ways in which nebulae and stars are different? a. stars can form inside a nebula but a nebula can never be produced by any star. b. a star always has a higher density than a nebula. c. stars can never form inside a nebula but a nebula can be produced by any star. d. a nebula always has a higher density than a star.
Answers: 3
You know the right answer?
A student prepared a stock solution by dissolving 20.0 g of NaOH in enough water to make 150. mL of...
Questions



Mathematics, 02.10.2020 09:01




Chemistry, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

Health, 02.10.2020 09:01


Biology, 02.10.2020 09:01

Mathematics, 02.10.2020 09:01

History, 02.10.2020 09:01


Mathematics, 02.10.2020 09:01



Advanced Placement (AP), 02.10.2020 09:01


History, 02.10.2020 09:01

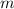 = Mass of sample =
= Mass of sample = 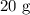
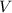 = Volume of solution =
= Volume of solution = 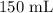
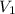 = Initial volume taken out of the stock solution =
= Initial volume taken out of the stock solution = 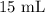
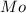 = Molar mass of NaOH =
= Molar mass of NaOH = 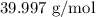
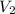 = Final volume of solution =
= Final volume of solution =