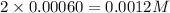Hydrogen sulfide decomposes according to the following reaction, for which Kc=9.30E-8 at 700 degrees Celsius. 2 H2S(g) --> 2 H2(g) + S2(g) If 0.29 moles of H2S is placed in a 3.0-L container, What is the equilibrium concentration of H2(g) at 700 degrees Celsius?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 13:00
6. using 3 – 4 sentences explain (in your own words) why water expands when it freezes? 7. using your knowledge of colligative properties explain whether sodium chloride or calcium chloride would be a more effective substance to melt the ice on a slick sidewalk. use 3 – 4 sentences in your explanation.
Answers: 1

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2


Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1
You know the right answer?
Hydrogen sulfide decomposes according to the following reaction, for which Kc=9.30E-8 at 700 degrees...
Questions


Mathematics, 14.04.2021 08:50

Biology, 14.04.2021 08:50


Mathematics, 14.04.2021 08:50

Mathematics, 14.04.2021 08:50

History, 14.04.2021 08:50


Arts, 14.04.2021 09:00








Arts, 14.04.2021 09:00


Mathematics, 14.04.2021 09:00

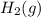 at 700 degrees Celsius is 0.0012 M
at 700 degrees Celsius is 0.0012 M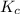
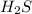 = 0.29 mole
= 0.29 mole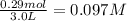
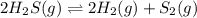
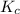 is written as:
is written as:![K_c=\frac{[H_2]^2\times [S_2]}{[H_2S]^2}](/tpl/images/0827/9052/13e21.png)
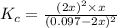
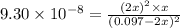
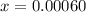
![[H_2]](/tpl/images/0827/9052/08a38.png) = 2x=
= 2x=