What makes balancing redox reactions different from balancing other
reactions?
A. Both the nu...

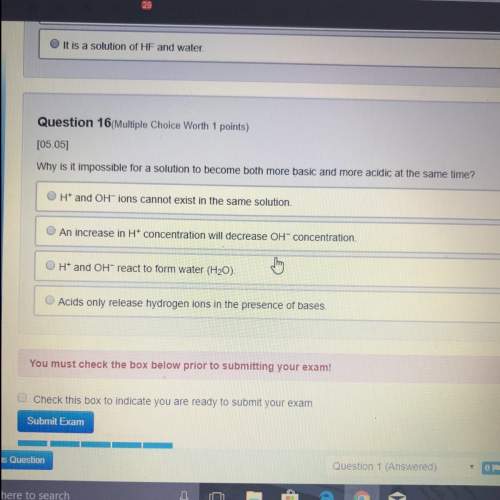
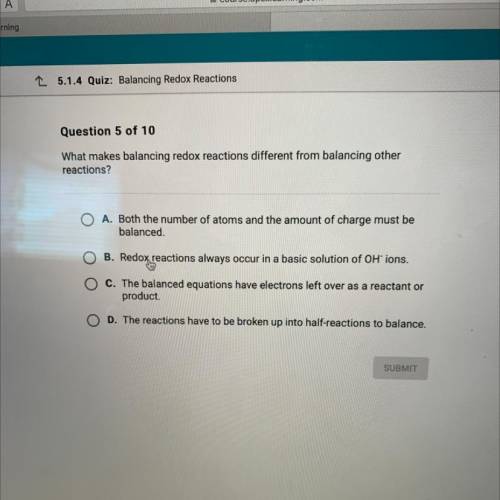
What makes balancing redox reactions different from balancing other
reactions?
A. Both the number of atoms and the amount of charge must be
balanced.
B. Redox reactions always occur in a basic solution of OH ions.
ChThe balanced equations have electrons left over as a reactant or
"product.
D. The reactions have to be broken up into half-reactions to balance.
SUBMIT

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The variability in marine salinity between habitats does not impact the fish living there. select the best answer from the choices provided t f
Answers: 1


Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 12:00
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1
You know the right answer?
Questions

Mathematics, 25.01.2021 07:30


English, 25.01.2021 07:30



Mathematics, 25.01.2021 07:30


English, 25.01.2021 07:30







Biology, 25.01.2021 07:30

History, 25.01.2021 07:30

Social Studies, 25.01.2021 07:30



Physics, 25.01.2021 07:30