Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 19:00
What information does a complete ionic equation give that the balanced equation doesn’t show?
Answers: 1

Chemistry, 22.06.2019 22:30
Which of the following is true about the speed of light? it depends on the wavelength.
Answers: 3

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
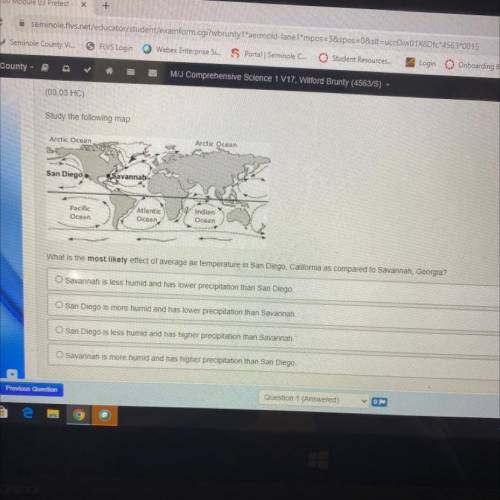
Study the following map:
What is the most likely effect of average air temperature in San Diego, Ca...
Questions

Mathematics, 01.08.2021 08:50


Health, 01.08.2021 08:50


Mathematics, 01.08.2021 08:50






Mathematics, 01.08.2021 08:50

Chemistry, 01.08.2021 08:50




Social Studies, 01.08.2021 09:00

English, 01.08.2021 09:00

Mathematics, 01.08.2021 09:00

Advanced Placement (AP), 01.08.2021 09:00