
Chemistry, 17.10.2020 21:01 aambert7256
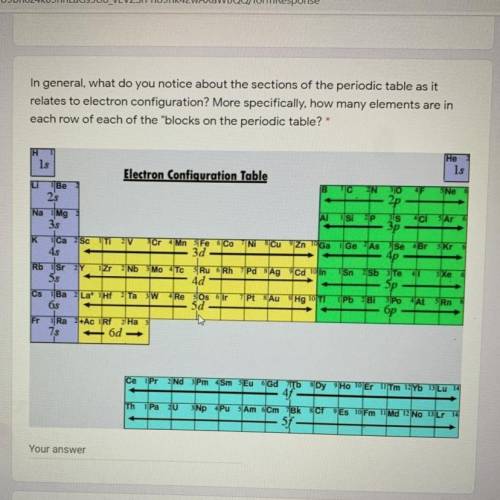
In general, what do you notice about the sections of the periodic table as it
relates to electron configuration? More specifically, how many elements are in
each row of each of the "blocks on the periodic table? *

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 21.06.2019 21:00
What pressure will be exerted by 0.675 moles of a gas at 25*c if it is in a 0.750-l container?
Answers: 1

Chemistry, 22.06.2019 00:30
Maria wants to determine which type of disinfectant kills the most bacteria. which of the following is the best way for maria to determine this? a. ask ten different companies that make disinfectants which type is best. b. put the same amount and species of bacteria on ten identical plates, and add ten different kinds of disinfectant to each plate. c. interview ten different people to determine which type of disinfectant they prefer. d. put the same amount and species of bacteria on ten identical plates, and add a different disinfectant to each plate.
Answers: 1

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1
You know the right answer?
In general, what do you notice about the sections of the periodic table as it
relates to electron c...
Questions

History, 16.02.2021 18:50

Mathematics, 16.02.2021 18:50



Mathematics, 16.02.2021 18:50

Mathematics, 16.02.2021 18:50

Advanced Placement (AP), 16.02.2021 18:50

English, 16.02.2021 18:50


Social Studies, 16.02.2021 19:00

Health, 16.02.2021 19:00


Mathematics, 16.02.2021 19:00




Biology, 16.02.2021 19:00

Spanish, 16.02.2021 19:00

History, 16.02.2021 19:00



