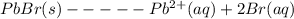Chemistry, 17.10.2020 16:01 ilovemeatballs5
Determine the value of the equilibrium constant (report your answer to three significant figures) for the following reaction if an equilibrium mixture contains 0.010 mol of solid PbBr2, and is 0.0100 M in Pb2+ ions and 0.0250 M in Br1- ions. Use the notation 4.31e-5 to indicate a number such as 4.31 x 10-5.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Given that 1 mi = 1760 yd, determine what conver- sion factor is appropriate to convert 1849 yd to miles; to convert 2.781 mi to yards.
Answers: 2

Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3

Chemistry, 22.06.2019 19:50
A2.5% (by mass) solution concentration signifies that there is a 2.5 % (by mass) solution concentration signifies that there is blank of solute in every 100 g of solution. of solute in every 100 g of solution
Answers: 3
You know the right answer?
Determine the value of the equilibrium constant (report your answer to three significant figures) fo...
Questions


Mathematics, 03.08.2019 01:50

Social Studies, 03.08.2019 01:50


Biology, 03.08.2019 01:50

Health, 03.08.2019 01:50

Computers and Technology, 03.08.2019 01:50

Geography, 03.08.2019 01:50

Geography, 03.08.2019 01:50

Geography, 03.08.2019 01:50


Biology, 03.08.2019 01:50


History, 03.08.2019 01:50

History, 03.08.2019 01:50

Mathematics, 03.08.2019 01:50

Mathematics, 03.08.2019 01:50


Chemistry, 03.08.2019 01:50

Mathematics, 03.08.2019 01:50