
Chemistry, 17.10.2020 14:01 brandicarney70p8jlsp
Please help asap !
3. Give the chemical equations for each single replacement reaction
that took place
4. Was Fe 3+ reduced? If so, what metal(s) acted as reducing agents?
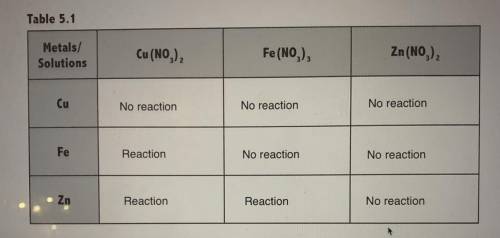

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1


Chemistry, 23.06.2019 14:30
William has eight more nickels than dimes in his pocket for a total of $2.50. which equation could be used to determine the number of x dimes in his pocket?
Answers: 1
You know the right answer?
Please help asap !
3. Give the chemical equations for each single replacement reaction
that t...
that t...
Questions

History, 24.11.2019 05:31

Biology, 24.11.2019 05:31

Mathematics, 24.11.2019 05:31



History, 24.11.2019 05:31


Biology, 24.11.2019 05:31

History, 24.11.2019 05:31

English, 24.11.2019 05:31


Mathematics, 24.11.2019 05:31


Mathematics, 24.11.2019 05:31






Biology, 24.11.2019 05:31



