
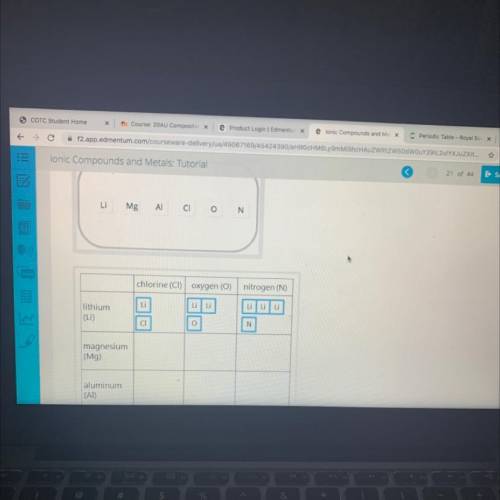
The ratio of the lons in an ionic compound depends on the charges of the ions. The amount of positive charge
must balance the amount of negative charge. For each compound, both the total positive charge and the total
negative charge will equal the LCM found in part B.
Determine the number of positive ions and the number of negative ions for each ionic compound in the table.
Use the lonic charges of each element you found in part A. The number of each ion times the charge on the lon
should equal the LCM determined for each cell in part B.
[# of ions of an element] * [lon charge) - least common multiple (LCM)
Drag each element's chemical symbol to the table to show how many lons are needed to balance the charges.
Each symbol will be used more than once. The first row is already completed for you.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 23.06.2019 02:00
What is the difference between a substance "getting wet" and "being dissolved" in a liquid at the particulate level?
Answers: 3

Chemistry, 23.06.2019 04:00
Which of these are physical changes in matter? check all that apply boiling water a pencil being sharpened exploding dynamite freezing water rotting cheese
Answers: 1
You know the right answer?
The ratio of the lons in an ionic compound depends on the charges of the ions. The amount of positiv...
Questions


Mathematics, 03.06.2020 14:02

Mathematics, 03.06.2020 14:02


Mathematics, 03.06.2020 14:02

Mathematics, 03.06.2020 14:02





Biology, 03.06.2020 14:02

Physics, 03.06.2020 14:02


Mathematics, 03.06.2020 14:02

History, 03.06.2020 14:02


Biology, 03.06.2020 14:02



Mathematics, 03.06.2020 14:02



