
Chemistry, 16.10.2020 20:01 hargunk329
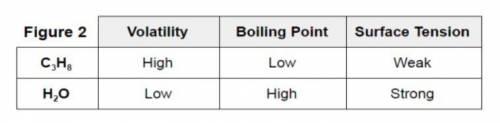
Consider the information provided in experiment #3 Figure 2. Three different variables are compared for C3H8 (propane) and H2O (water). The strength of the intermolecular forces are responsible for the behavior of the two compounds.
Identify the intermolecular force for the two compounds.
Which one is stronger?
Explain how the strength of intermolecular forces plays a role in at least two of the properties (volatility, boiling point, or surface tension) for C3H8 (propane) and H2O (water).
Cite evidence from the data to explain your reasoning.
Experiment #3: The student follows study #2 with a comparison of propane (C3H8) and water. Volatility is the tendency for a substance to vaporize and it was found that propane vaporizes quickly while water vaporized slowly. Surface tension is a physical property equal to the amount of force per unit area necessary to expand the surface of a liquid. It is this phenomenon which allows a water strider or lizard to run across the surface of water.
100 points

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
How do scientists think that gravity affected the formation of our solar system?
Answers: 1

Chemistry, 22.06.2019 09:20
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 22.06.2019 16:00
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3
You know the right answer?
Consider the information provided in experiment #3 Figure 2. Three different variables are compared...
Questions

Mathematics, 12.11.2020 22:50

History, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50



English, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50

Advanced Placement (AP), 12.11.2020 22:50

Mathematics, 12.11.2020 22:50

Health, 12.11.2020 22:50

Computers and Technology, 12.11.2020 22:50


Mathematics, 12.11.2020 22:50

Social Studies, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50


Mathematics, 12.11.2020 22:50

English, 12.11.2020 22:50




