
Chemistry, 16.10.2020 08:01 thelonewolf5020
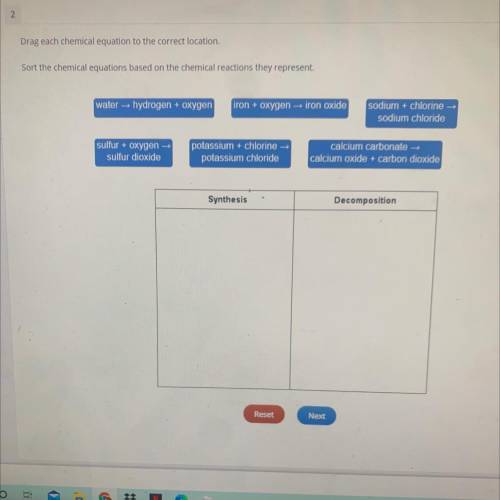
Drag each chemical equation to the correct location.
Sort the chemical equations based on the chemical reactions they represent.
water hydrogen + oxygen
iron + oxygen iron oxide
sodium + chlorine
sodium chloride
sulfur + oxygen
sulfur dioxide
potassium + chlorine-
potassium chloride
calcium carbonate
calcium oxide + carbon dioxide
Synthesis
Decomposition

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Transportation is the largest single source of air pollution in the united states. air pollution can harm the environment and human health. which technology could offer a solution to this problem? mufflers that reduce noise motors that run on electricity tires that improve gas mileage
Answers: 3

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1
You know the right answer?
Drag each chemical equation to the correct location.
Sort the chemical equations based on the chemi...
Questions

History, 06.11.2020 02:00


History, 06.11.2020 02:00



Mathematics, 06.11.2020 02:00

Arts, 06.11.2020 02:00

Chemistry, 06.11.2020 02:00


Mathematics, 06.11.2020 02:00


English, 06.11.2020 02:00




Mathematics, 06.11.2020 02:00

Mathematics, 06.11.2020 02:00


Biology, 06.11.2020 02:00




