Chemistry, 16.10.2020 08:01 jamccoy3335
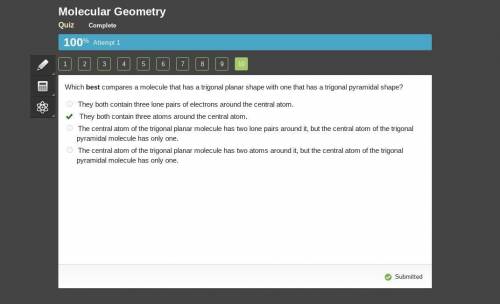
Which best compares a molecule that has a trigonal planar shape with one that has a trigonal pyramidal shape?
O They both contain three lone pairs of electrons around the central atom.
O They both contain three atoms around the central atom.
O The central atom of the trigonal planar molecule has two lone pairs around it, but the central atom of the trigonal
pyramidal molecule has only one.
O The central atom of the trigonal planar molecule has two atoms around it, but the central atom of the trigonal
pyramidal molecule has only one.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1

Chemistry, 22.06.2019 16:00
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2

Chemistry, 22.06.2019 23:30
Match each statement with the state of matter it describes
Answers: 3

Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2
You know the right answer?
Which best compares a molecule that has a trigonal planar shape with one that has a trigonal pyramid...
Questions

History, 02.07.2019 12:20


Mathematics, 02.07.2019 12:20


Health, 02.07.2019 12:20



Mathematics, 02.07.2019 12:20


History, 02.07.2019 12:20

Arts, 02.07.2019 12:20

English, 02.07.2019 12:20

Mathematics, 02.07.2019 12:20



Mathematics, 02.07.2019 12:30


Social Studies, 02.07.2019 12:30


Mathematics, 02.07.2019 12:30