
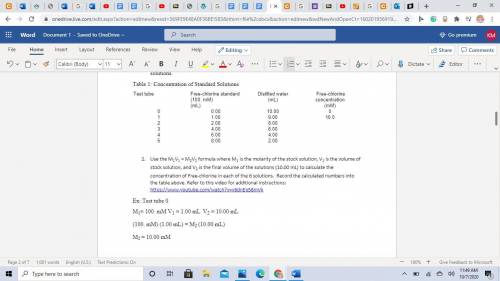
A 100mM/L solution of Free-chlorine stock solution was used to make up 6 standard solutions (known concentration). See the table below with gives the test tube number, the amount of stock solution used and the amount of distilled water used to make up these solutions.
Table 1: Concentration of Standard Solutions
Test tube
Free-chlorine standard (100. mM)
(mL)
Distilled water
(mL)
Free-chlorine concentration
(mM)
0
0.00
10.00
0
1
1.00
9.00
10.0
2
2.00
8.00
3
4.00
6.00
4
6.00
4.00
5
8.00
2.00
Use the M1V1 = M2V2 formula where M1 is the molarity of the stock solution, V1 is the volume of stock solution, and V2 is the final volume of the solutions (10.00 mL) to calculate the concentration of Free-chlorine in each of the 6 solutions. Record the calculated numbers into the table above. Ex: Test tube 0
M1= 100. mM V1 = 1.00 mL V2 = 10.00 mL
(100. mM) (1.00 mL) = M2 (10.00 mL)
M2 = 10.00 mM
(there's also a screenshot of the problem if that's easier)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
In the millikan oil drop experiment they determined that every drop had a charge which was a while number multiple of -1.60x10^-19. if a drop has a total charge of -9.60x10^-19 then how many excess electrons are contained within the drop?
Answers: 2


Chemistry, 23.06.2019 07:00
What are the trends and exceptions to the trends in electron affinity?
Answers: 1

Chemistry, 23.06.2019 14:00
How is the electron sea model of metallic bonding different from the band theory? how are they the same? give at least one similarity and one difference between the models
Answers: 2
You know the right answer?
A 100mM/L solution of Free-chlorine stock solution was used to make up 6 standard solutions (known c...
Questions




Mathematics, 04.03.2021 02:00

Mathematics, 04.03.2021 02:00

Mathematics, 04.03.2021 02:00

Mathematics, 04.03.2021 02:00

Mathematics, 04.03.2021 02:00

History, 04.03.2021 02:00




Mathematics, 04.03.2021 02:00




Mathematics, 04.03.2021 02:00

Mathematics, 04.03.2021 02:00

Mathematics, 04.03.2021 02:00

Computers and Technology, 04.03.2021 02:00



