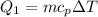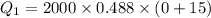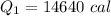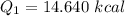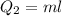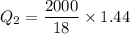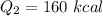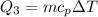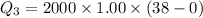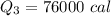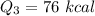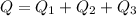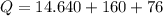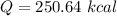Cold-winter survival experts do not recommend eating snow for hydration. One reason for this is the energy it takes to increase temperature of the snow, melt it, and then increase the temperature of the resulting water to body temperature. Calculate the total amount of heat to convert 2.000 kg of snow at -15.00 0C to liquid water at 38.00 0C.
POTENTIALLY HELPFUL INFORMATION:
The heat of fusion for water is 1.44 kcal/mol.
The specific heat capacity of solid water is 0.488 cal/g0C.
The specific heat capacity of liquid water is 1.00 cal/g0C.
The freezing point of water is 00C (also known as melting point).

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2


Chemistry, 23.06.2019 02:00
Which would freeze at a higher temperature: the great salt lake or lake tahoe? a. lake tahoe would freeze at a higher temperature. b. the great salt lake would freeze at a higher temperature. c. both lakes would freeze at the same temperature.
Answers: 2
You know the right answer?
Cold-winter survival experts do not recommend eating snow for hydration. One reason for this is the...
Questions

English, 12.02.2021 22:40



Mathematics, 12.02.2021 22:40

Arts, 12.02.2021 22:40





Arts, 12.02.2021 22:40

Chemistry, 12.02.2021 22:40

English, 12.02.2021 22:40





Mathematics, 12.02.2021 22:40


Mathematics, 12.02.2021 22:40