
Chemistry, 02.10.2020 19:01 jtswagg6634
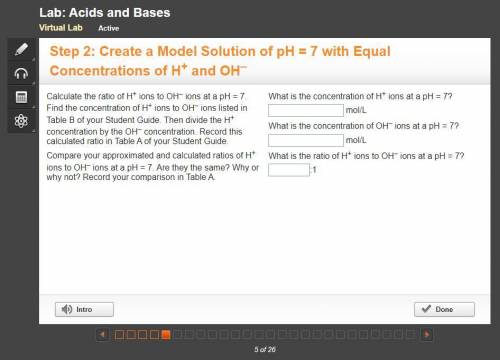
10 PTS FOR CORRECT ANSWER Calculate the ratio of H+ ions to OH– ions at a pH = 7. Find the concentration of H+ ions to OH– ions listed in Table B of your Student Guide. Then divide the H+ concentration by the OH– concentration. Record this calculated ratio in Table A of your Student Guide.
Compare your approximated and calculated ratios of H+ ions to OH– ions at a pH = 7. Are they the same? Why or why not? Record your comparison in Table A.
What is the concentration of H+ ions at a pH = 7?
mol/L
What is the concentration of OH– ions at a pH = 7?
mol/L
What is the ratio of H+ ions to OH– ions at a pH = 7?
:1

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2

Chemistry, 22.06.2019 23:30
What are the similarities between compounds and mixtures?
Answers: 3
You know the right answer?
10 PTS FOR CORRECT ANSWER Calculate the ratio of H+ ions to OH– ions at a pH = 7. Find the concentra...
Questions






Mathematics, 18.07.2019 14:40

Social Studies, 18.07.2019 14:40




Mathematics, 18.07.2019 14:40

History, 18.07.2019 14:40



History, 18.07.2019 14:40

History, 18.07.2019 14:40

History, 18.07.2019 14:40






