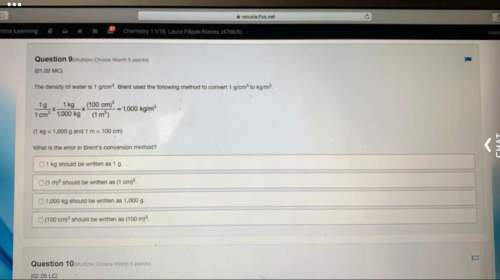
Chemistry, 28.09.2020 05:01 garrettadkins2002
How small is an atom. Write down the calculation.
The diameter of a US penny is 19 mm. The diameter of a silver atom, by comparison, is only 2.88 *10 -10mm.
How many silver atoms could be arranged side by side in a straight line at diameter of a penny (d penny)? 10 min
d penny =19mm I # Ag = d penny /d Ag atom I # Ag=19mm /2.88 *10 -10mm = 6.6*106 atoms
d Ag atom =2.88 *10 -10mm [ 6 600 000 atoms]
# Ag atoms--->? Atoms

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 22.06.2019 13:00
The number of neutrons is equal to the atomic number minus the atomic mass. a. true b. false
Answers: 2


Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2
You know the right answer?
How small is an atom. Write down the calculation.
The diameter of a US penny is 19 mm. The diameter...
Questions


Biology, 10.05.2021 19:10



Physics, 10.05.2021 19:10

Engineering, 10.05.2021 19:10

Mathematics, 10.05.2021 19:10


Mathematics, 10.05.2021 19:10

Mathematics, 10.05.2021 19:10

Mathematics, 10.05.2021 19:10


History, 10.05.2021 19:10


Advanced Placement (AP), 10.05.2021 19:10

Mathematics, 10.05.2021 19:10


Mathematics, 10.05.2021 19:10

Mathematics, 10.05.2021 19:10