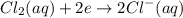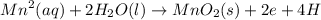Chemistry, 23.09.2020 08:01 jhenderson2024
A galvanic cell is powered by the following redox reaction: (aq) (aq) (aq)(s) (l) (l) Answer the following questions about this cell. If you need any electrochemical data, be sure you get it from the ALEKS Data tab. Write a balanced equation for the half-reaction that takes place at the cathode. Write a balanced equation for the half-reaction that takes place at the anode. Calculate the cell voltage under standard conditions. Round your answer to decimal places.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2


Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1

You know the right answer?
A galvanic cell is powered by the following redox reaction: (aq) (aq) (aq)(s) (l) (l) Answer the fol...
Questions


Mathematics, 08.06.2021 17:20




Mathematics, 08.06.2021 17:20

History, 08.06.2021 17:20


Biology, 08.06.2021 17:20



Business, 08.06.2021 17:20