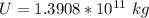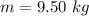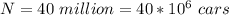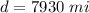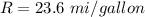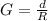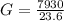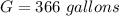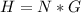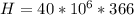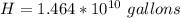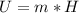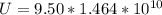One gallon of gasoline in an automobiles engine produces on average 9.50 kg of carbon dioxide, which is a greenhouse gas; that is, it promotes the warming of Earth's atmosphere. Calculate the annual production of carbon dioxide in kilograms if there are exactly 40.0 million cars in the United States and each car covers a distance of 7930 mi at a consumption rate of 23.6 miles per gallon. Enter your answer in scientific notation.

Answers: 1


Another question on Chemistry


Chemistry, 23.06.2019 00:00
Exit what is the density of an object having a mass of 5.0 g and a volume of 45.0 cm3?
Answers: 1

Chemistry, 23.06.2019 08:00
Amechanical wave that transports a lot of energy will have a
Answers: 2

Chemistry, 23.06.2019 14:30
Which statement best identifies the process shown? the process must be fusion because energy is released. a.the process must be fusion because a heavier nucleus forms from smaller nuclei. b.the process must be fission because a large nucleus breaks into smaller nuclei. c.the process must be fission because neutrons are formed.
Answers: 1
You know the right answer?
One gallon of gasoline in an automobiles engine produces on average 9.50 kg of carbon dioxide, which...
Questions


History, 28.09.2019 19:30


Social Studies, 28.09.2019 19:30

History, 28.09.2019 19:30



Mathematics, 28.09.2019 19:30

Mathematics, 28.09.2019 19:30


Biology, 28.09.2019 19:30

Mathematics, 28.09.2019 19:30

Chemistry, 28.09.2019 19:30


Chemistry, 28.09.2019 19:30

Mathematics, 28.09.2019 19:30

Biology, 28.09.2019 19:30

Mathematics, 28.09.2019 19:30


History, 28.09.2019 19:30