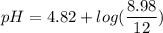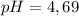Chemistry, 04.09.2020 21:01 tinytoonjr6901
2.643 grams of potassium butanoate (KCH3(CH2)2CO2 ) is fully dissolved in 50.00 mL of water, which is carefully transferred to a conical flask. Then 100.00 mL of 0.120 M HCℓ is added dropwise to this solution from a burette. Given: Ka(butanoic acid) = 1.5 × 1O−5 . 2.1 Showing all your calculations and reasoning, determine the pH of the solution that results after the addition of all the acid mentioned above.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:00
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1

Chemistry, 21.06.2019 19:30
Determine the number o moles of ions/atoms/particle in the following: 2.50 miles of k2s (let me know how to do)
Answers: 1

Chemistry, 21.06.2019 23:00
Matches the chemical name of each oxide of phosphorus to its chemical formula
Answers: 2

Chemistry, 22.06.2019 03:00
Match term definition ellipse a) diagonal cross section of a cylinder circle b) diagonal cross section through the widest part of a sphere sphere c) cross section parallel to the base of a cone great circle d) shape created when a semi-circle is rotated around the y-axis triangle e) perpendicular cross section of a cone
Answers: 1
You know the right answer?
2.643 grams of potassium butanoate (KCH3(CH2)2CO2 ) is fully dissolved in 50.00 mL of water, which i...
Questions


Arts, 24.11.2019 23:31



Biology, 24.11.2019 23:31





Mathematics, 24.11.2019 23:31

English, 24.11.2019 23:31


Mathematics, 24.11.2019 23:31

Mathematics, 24.11.2019 23:31

Geography, 24.11.2019 23:31

Mathematics, 24.11.2019 23:31

Health, 24.11.2019 23:31



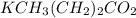 is a basic salt.
is a basic salt.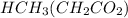 would become HY.
would become HY.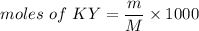
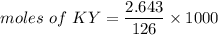
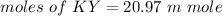
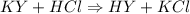
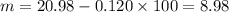
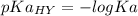
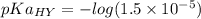
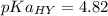
![pH=pKa+log(\dfrac{[KY]}{[KH]})](/tpl/images/0742/1299/49770.png)