Chemistry, 02.09.2020 23:01 justhereforanswers13
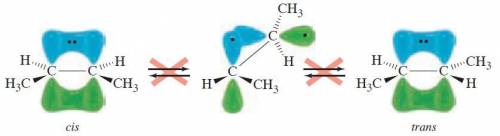
Rotation about a carbon-carbon double bond does not readily occur because: .1) the overlap of the p orbitals of the carbon-carbon π bond would be lost2) the double bond is much shorter and therefore more difficult to rotate3) the overlap of the sp2 orbitals of the carbon-carbon σ bond would be lost4) the double bond is much stronger and therefore more difficult to rotate

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 07:30
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н,о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 11:50
The chemical bond connecting one nucleotide with the next one along the nucleic acid chain is called a
Answers: 3
You know the right answer?
Rotation about a carbon-carbon double bond does not readily occur because: .1) the overlap of the p...
Questions


English, 07.10.2019 20:00


History, 07.10.2019 20:00

English, 07.10.2019 20:00

Computers and Technology, 07.10.2019 20:00

Health, 07.10.2019 20:00


Mathematics, 07.10.2019 20:00


History, 07.10.2019 20:00

Computers and Technology, 07.10.2019 20:00






Computers and Technology, 07.10.2019 20:00