Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:00
What is the nature of the ca-cl bond in a molecule of calcium chloride (cacl2) if the electronegativity value of calcium is 1.0 and that of chlorine is 3.16?
Answers: 1

Chemistry, 21.06.2019 18:00
Construct the hypothetical phase diagram for metals a and b between room temperature (20c) and 700c, given the following information: * the melting temperature of metal a is 480c. • the maximum solubility of b in a is 4 wt% b, which occurs at 420c. • the solubility of b in a at room temperature is 0 wt% b. • one eutectic occurs at 420c and 18 wt% b–82 wt% a. • a second eutectic occurs at 475c and 42 wt% b–58 wt% a. • the intermetallic compound ab exists at a composition of 30 wt% b–70 wt% a, and melts congruently at 525c.• the melting temperature of metal b is 600c. • the maximum solubility of a in b is 13 wt% a, which occurs at 475c. • the solubility of a in b at room temperature is 3 wt% a.
Answers: 1

Chemistry, 22.06.2019 07:10
Which of these conditions most likely produces an unstable isotope?
Answers: 2

Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1
You know the right answer?
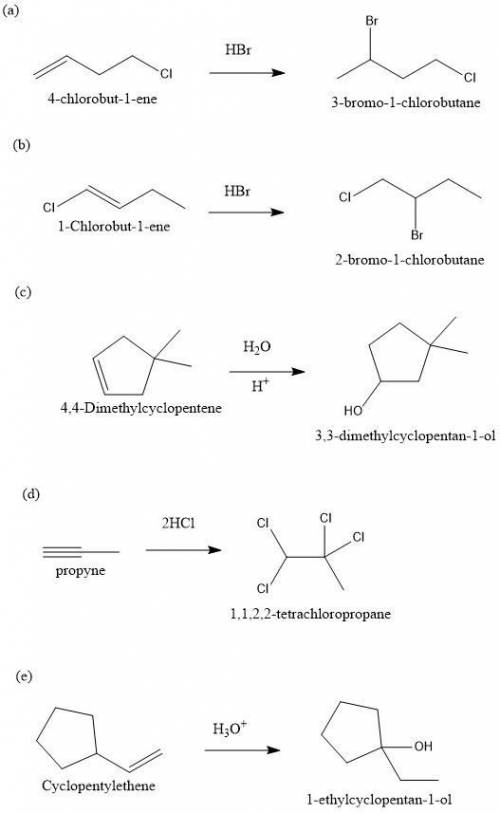
Predict the major product(s) for each of the following reactions. H3O (a) 4-Chlorobut-1-ene HBr (b)...
Questions

History, 19.02.2021 21:50

Mathematics, 19.02.2021 21:50

History, 19.02.2021 21:50




Mathematics, 19.02.2021 21:50

Mathematics, 19.02.2021 21:50


History, 19.02.2021 21:50


History, 19.02.2021 21:50

Mathematics, 19.02.2021 21:50

Mathematics, 19.02.2021 21:50



History, 19.02.2021 21:50



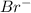 " will be added to the most substituted carbon. In this case carbon 3 to produce 3-bromo-1-chlorobutane.
" will be added to the most substituted carbon. In this case carbon 3 to produce 3-bromo-1-chlorobutane.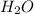 " will be added to the most substituted carbon. In this case carbon 3 to produce 3,3-dimethylcyclopentan-1-ol.
" will be added to the most substituted carbon. In this case carbon 3 to produce 3,3-dimethylcyclopentan-1-ol.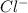 " will be added to the most substituted carbon, but we have 2 moles of the nucleophile, so would be added 2 times and we will have as product 1,1,2,2-tetrachloropropane.
" will be added to the most substituted carbon, but we have 2 moles of the nucleophile, so would be added 2 times and we will have as product 1,1,2,2-tetrachloropropane.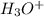 " will be added to the most substituted carbon. But in this case, the carbon cation would be produced in carbon 1 of the ethene. So, we can have a hydride shift to produce a tertiary carbocation. With this in mind the nucleohile will be addde to this tertiary carbocation and we will have 1-ethylcyclopentan-1-ol.
" will be added to the most substituted carbon. But in this case, the carbon cation would be produced in carbon 1 of the ethene. So, we can have a hydride shift to produce a tertiary carbocation. With this in mind the nucleohile will be addde to this tertiary carbocation and we will have 1-ethylcyclopentan-1-ol.