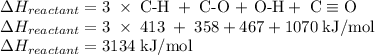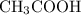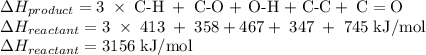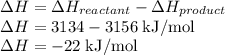Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 16:50
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 23.06.2019 02:30
Apound is approximately 0.45 kilogram. a persons weighs 87 kilograms. what is the persons’s weight, in pounds, when expressed to the correct number of significant figures
Answers: 1
You know the right answer?
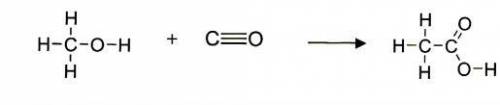
An important industrial route to extremely pure acetic acid is the reaction of methanol with carbon...
Questions



Mathematics, 19.12.2021 18:00


Chemistry, 19.12.2021 18:00

Mathematics, 19.12.2021 18:00

History, 19.12.2021 18:00


Biology, 19.12.2021 18:00





Computers and Technology, 19.12.2021 18:00


Mathematics, 19.12.2021 18:00

Social Studies, 19.12.2021 18:00



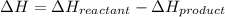
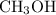 and
and  .
.