
Chemistry, 28.08.2020 20:01 eshaesmot12345
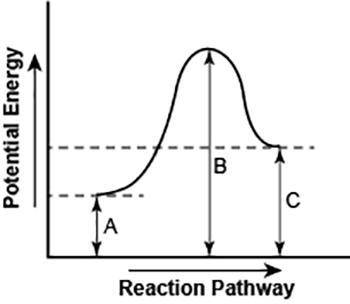
Part 1: Describe how you can determine the total change in enthalpy and activation energy from the diagram, and if each is positive or negative. Part 2: Describe how the curve will look if the reaction was exothermic. Be sure to mention changes in the potential energies of the reactants and products and the sign changes of the enthalpy.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Asample contains 16.75 g of the radioisotope u-236 and 50.25 g of its daughter isotope, th-232. how long did it take for decay to take place if one half-life of u-236 is 23 million years? 46 million years 69 million years 92 million years 115 million years
Answers: 3

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3

Chemistry, 21.06.2019 23:00
What is the formula that this ionic compounds could form sr2+p3-o2-
Answers: 3

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1
You know the right answer?
Part 1: Describe how you can determine the total change in enthalpy and activation energy from the d...
Questions






SAT, 14.12.2020 19:40



Mathematics, 14.12.2020 19:40





Advanced Placement (AP), 14.12.2020 19:40


Mathematics, 14.12.2020 19:40



Mathematics, 14.12.2020 19:40



