
Chemistry, 27.08.2020 21:01 oksanabkrot
A reaction between 7.0 g of copper(II) oxide and 50 mL of 0.20 M nitric acid produces
copper(II) nitrate, Cu(NO3)2 and water.
(c) Determine the limiting reactant.
(d) Calculate the mass of excess reactant after the reaction.
(ANS: 6.6068g)
(e) Determine the percentage yield if the actual mass of copper (II) nitrate obtained from
the reaction is 0.85 g.
(ANS: 90.64%)
How to get the mass of HNO3 from here? I only managed to get mass of NO3 based on the molarity formula. thanks!

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:30
Water molecules have a strong attraction to each other because of hydrogen bonding, allowing water to move against gravity up a plant's stem through capillary action. true false
Answers: 2

Chemistry, 22.06.2019 02:30
Needthe meter is the standard unit for: 1) height 2) length 3) weight 4) mass
Answers: 3

Chemistry, 22.06.2019 05:00
When you mate two plants together the terms is called? answer it fast as possible plz! i have a test tomorrow!
Answers: 1

Chemistry, 22.06.2019 14:30
The three types is stress that act on earths rocks are compression, tension, and
Answers: 1
You know the right answer?
A reaction between 7.0 g of copper(II) oxide and 50 mL of 0.20 M nitric acid produces
copper(II) ni...
Questions




English, 12.05.2021 20:30

Biology, 12.05.2021 20:30

Arts, 12.05.2021 20:30


Health, 12.05.2021 20:30

Social Studies, 12.05.2021 20:30

Chemistry, 12.05.2021 20:30

Mathematics, 12.05.2021 20:30

Mathematics, 12.05.2021 20:30


Mathematics, 12.05.2021 20:30

Mathematics, 12.05.2021 20:30



Mathematics, 12.05.2021 20:30


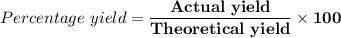
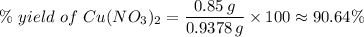
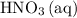 is the limiting reactant.
is the limiting reactant.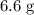 of
of 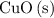 will be in excess.
will be in excess.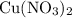 is approximately
is approximately 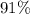 . (Rounded to two significant figures, as in other quantities in the question.)
. (Rounded to two significant figures, as in other quantities in the question.)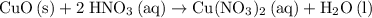 .
.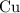 :
: 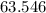 .
. :
:  .
. :
: 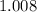 .
.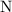 :
: 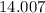 .
.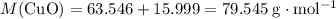 .
.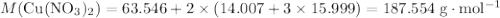 .Limiting Reactant
.Limiting Reactant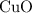 and
and 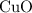 is the limiting one. In other words, assume that all the
is the limiting one. In other words, assume that all the 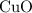 is consumed before
is consumed before 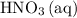 was.
was.  would be required to convert all that
would be required to convert all that 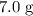 of
of 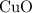 to
to  of
of 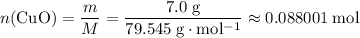 .
.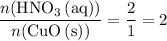 .
.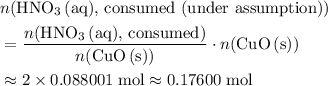 .
. 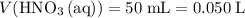 .
.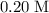 solution:
solution: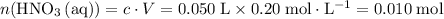 .
.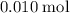 of
of 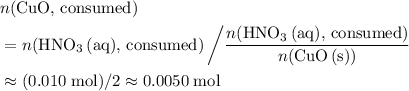 .
. . Therefore, the mass of that
. Therefore, the mass of that 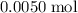 formula units of
formula units of 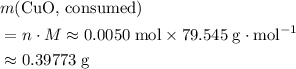 .
.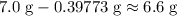 of
of 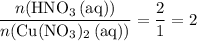 .
.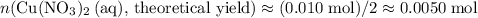 .
.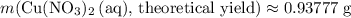 .
.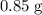 :
: .
.

