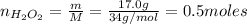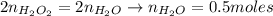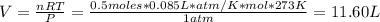Chemistry, 27.08.2020 04:01 kmshacklette9879
Hydrogen peroxide (H2O2, 34 g/mol) decomposes into water vapor and oxygen gas. How many liters of water vapor are produced from the decomposition of 17.0 g of H2O2 at STP?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 00:00
Explain which group an element with the electron configuration 1s2 2s2 2p6 3s2 3p6 3d1 4s2 belongs to.
Answers: 3

Chemistry, 22.06.2019 01:00
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 22.06.2019 15:10
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1
You know the right answer?
Hydrogen peroxide (H2O2, 34 g/mol) decomposes into water vapor and oxygen gas. How many liters of wa...
Questions




History, 12.02.2020 21:43


Mathematics, 12.02.2020 21:43

Mathematics, 12.02.2020 21:43

History, 12.02.2020 21:43

History, 12.02.2020 21:43


Mathematics, 12.02.2020 21:43

Engineering, 12.02.2020 21:44




Physics, 12.02.2020 21:44

Biology, 12.02.2020 21:44

Biology, 12.02.2020 21:44