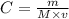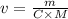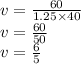Chemistry, 23.08.2020 17:01 matthewquattlebum
What volume of 1.25 M sodium hydroxide (NaOH) solution can be prepared using 60.0 g of sodium hydroxide? (The mass of one mole of NaOH is 40.0 g.)
0.667 L
0.833 L
1.20 L
1.88L

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 23.06.2019 01:00
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 03:20
High-pressure liquid chromatography (hplc) is a method used in chemistry and biochemistry to purify chemical substances. the pressures used in this procedure range from around 500 kilopascals (500,000 pa) to about 60,000 kpa (60,000,000 pa). it is often convenient to know the pressure in torr. if an hplc procedure is running at a pressure of 1.03×108 pa , what is its running pressure in torr?
Answers: 3

Chemistry, 23.06.2019 05:00
How is electrolysis most commonly used to produce an energy source? a - splitting water molecules produces oxygen, which organisms breathe to fuel their bodies. b - splitting water molecules produces hydrogen gas, which is used to power machines through hydrogen fuel cells. c - splitting carbon dioxide molecules produces coal, a form of carbon that can be burned to produce heat. d - splitting carbon dioxide molecules produces natural gas, which can be burned to generate electricity in power plants.
Answers: 1
You know the right answer?
What volume of 1.25 M sodium hydroxide (NaOH) solution can be prepared using 60.0 g of sodium hydrox...
Questions


Biology, 17.01.2020 00:31


Mathematics, 17.01.2020 00:31


English, 17.01.2020 00:31

Mathematics, 17.01.2020 00:31


English, 17.01.2020 00:31

Computers and Technology, 17.01.2020 00:31






Mathematics, 17.01.2020 00:31