

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 15:00
Many ionic compounds and a few highly polar covalent compounds are because they completely ionize in water to create a solution filled with charged ions that can conduct an electric current.
Answers: 1

Chemistry, 22.06.2019 17:00
Astable electron arrangement for an atom is one that does not easily change. how is this arrangement arrived at? a. valence electrons are transferred or shared to create a full outer shell of electrons. b. valence electrons are discarded into space to create a full outer shell of electrons. c. protons (positive charge) pair with valence electrons (negative charge) to create a strong bond. d. outer shells with valence electrons are transferred or shared.
Answers: 2
You know the right answer?
A 0.010 M aqueous solution of a weak acid HA has a pH of 4.0. What is the degree of ionization of HA...
Questions

Mathematics, 03.02.2022 20:00

Mathematics, 03.02.2022 20:00

Computers and Technology, 03.02.2022 20:10

Chemistry, 03.02.2022 20:10


Biology, 03.02.2022 20:10



Social Studies, 03.02.2022 20:10

Health, 03.02.2022 20:10

Mathematics, 03.02.2022 20:10





Arts, 03.02.2022 20:10

Mathematics, 03.02.2022 20:20



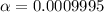
![pH=-log([H^+]})](/tpl/images/0726/6480/27fdb.png)
![[H^+]=10^{-pH}=10^{-4.0}=1x10^{-4}M](/tpl/images/0726/6480/17ea8.png)
 ) is computed as:
) is computed as:
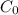 is 0.010 M and the acid dissociation constant is:
is 0.010 M and the acid dissociation constant is:



