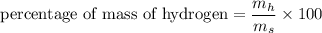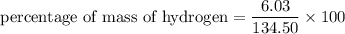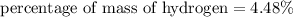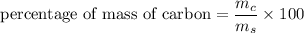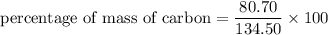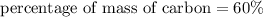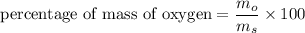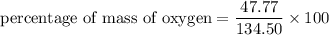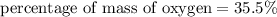Answers: 2


Another question on Chemistry


Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 21.06.2019 23:00
Ahypothrticalax type of ceramic material is known to have a density of 2.10 g/cm3 and a unit cell of cubic symmetry with a cell edge length of 0.57 nm. the atomic weights of the a and x elements are 28.5and 30.0 g/mol, respectively. on the basis of this information, which of the following crystal structures is (are) possible for this material: sodium chloride, cesium chloride, or zinc blende
Answers: 1

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1
You know the right answer?
3.A 134.50 g sample of aspirin is made up of 6.03 g of hydrogen, 80.70 g of carbon, and 47.77 g of o...
Questions





Chemistry, 05.03.2021 03:20

SAT, 05.03.2021 03:20

Mathematics, 05.03.2021 03:20

Physics, 05.03.2021 03:20

Arts, 05.03.2021 03:20


Mathematics, 05.03.2021 03:20



Mathematics, 05.03.2021 03:20




Mathematics, 05.03.2021 03:20