
Chemistry, 18.08.2020 17:01 cluchmasters5634
Consider the following reaction at equilibrium. What effect will increasing the pressure of the reaction mixture have on the system?
CuS(s) + O2(g) ? Cu(s) + SO2(g)
Consider the following reaction at equilibrium. What effect will increasing the pressure of the reaction mixture have on the system?
CuS(s) + O2(g) ? Cu(s) + SO2(g)
a) The equilibrium constant will increase.
b) The reaction will shift to the left in the direction of reactants.
c) The reaction will shift to the right in the direction of products.
d) No effect will be observed.
e) The equilibrium constant will decrease.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3



Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
Consider the following reaction at equilibrium. What effect will increasing the pressure of the reac...
Questions

Mathematics, 24.01.2021 23:20


Mathematics, 24.01.2021 23:20

English, 24.01.2021 23:20

Mathematics, 24.01.2021 23:20


Mathematics, 24.01.2021 23:20


Mathematics, 24.01.2021 23:20

Computers and Technology, 24.01.2021 23:20

Computers and Technology, 24.01.2021 23:20


Mathematics, 24.01.2021 23:20

English, 24.01.2021 23:20



Mathematics, 24.01.2021 23:20

Mathematics, 24.01.2021 23:20


Mathematics, 24.01.2021 23:20

![Q = \displaystyle \frac{[\mathrm{SO_2\, (g)}]}{[\mathrm{O_2\, (g)}]}](/tpl/images/0723/8610/a7d3d.png) .
.![[\mathrm{SO_2\, (g)}]](/tpl/images/0723/8610/2691f.png) and
and ![[\mathrm{O_2\, (g)}]](/tpl/images/0723/8610/46794.png) will increase if the pressure is increased through compression. However, because
will increase if the pressure is increased through compression. However, because 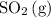 and
and 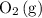 have the same coefficients in the equation, their concentrations are raised to the same power in the equilibrium quotient
have the same coefficients in the equation, their concentrations are raised to the same power in the equilibrium quotient 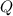 .
. 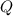 . If the system was already at equilibrium, it will continue to be at an equilibrium even after the change to its pressure. Therefore, no overall effect on the equilibrium position should be visible.
. If the system was already at equilibrium, it will continue to be at an equilibrium even after the change to its pressure. Therefore, no overall effect on the equilibrium position should be visible.

