Chemistry, 19.08.2020 03:01 school4life110
A buffer solution is 0.413 M in HF and 0.237 M in KF. If Ka for HF is 7.2×10-4, what is the pH of this buffer solution?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Each of the following compounds contains a metal that can exhibit more than one ionic charge. provide systematic names for each of these compounds. (a) cr(clo3)6 (b) mo(cn)6 (c) cr2(so3)3 (d) v(clo2)2 (e) v(cn)5 (f) os(clo2)4
Answers: 3


Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2
You know the right answer?
A buffer solution is 0.413 M in HF and 0.237 M in KF. If Ka for HF is 7.2×10-4, what is the pH of th...
Questions




Mathematics, 14.12.2021 02:10


English, 14.12.2021 02:10

SAT, 14.12.2021 02:10



Biology, 14.12.2021 02:10


Mathematics, 14.12.2021 02:10


Mathematics, 14.12.2021 02:10

Mathematics, 14.12.2021 02:10





Social Studies, 14.12.2021 02:10

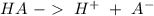
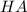 is the acid and
is the acid and 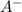 is the base. Additionally, the calculation of the pH of any buffer system can be made with the Henderson-Hasselbach equation:
is the base. Additionally, the calculation of the pH of any buffer system can be made with the Henderson-Hasselbach equation:![pH=pKa~+~Log\frac{[A^-]}{[HA]}](/tpl/images/0724/4288/665aa.png)
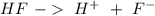
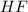 with a concentration of 0.413 M and the base is
with a concentration of 0.413 M and the base is 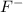 with a concentration of 0.237 M. We can calculate the pKa value if we do the "-Log Ka", so:
with a concentration of 0.237 M. We can calculate the pKa value if we do the "-Log Ka", so: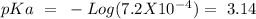
![pH=~3.14~+~Log(\frac{[0.237~M]}{[0.413~M]})~=~2.90](/tpl/images/0724/4288/bcf85.png)