Chemistry, 18.08.2020 22:01 allisongallion23
The standard free energy change for a reaction in an electrolytic cell is always:
a. Positive
b. Negative
c. Zero
d. Impossible to determine

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 07:00
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1
You know the right answer?
The standard free energy change for a reaction in an electrolytic cell is always:
a. Positive
Questions

Mathematics, 07.05.2021 05:00

Mathematics, 07.05.2021 05:00

English, 07.05.2021 05:00


Chemistry, 07.05.2021 05:00

Physics, 07.05.2021 05:00



Computers and Technology, 07.05.2021 05:00




Mathematics, 07.05.2021 05:00


Chemistry, 07.05.2021 05:00


History, 07.05.2021 05:00


Mathematics, 07.05.2021 05:00

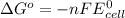
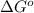 = standard free energy change
= standard free energy change 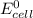 = standard emf
= standard emf