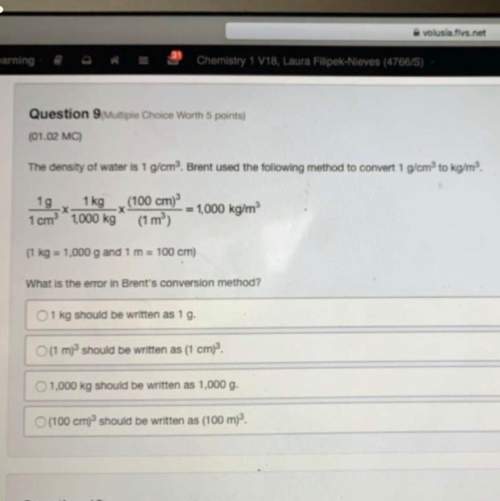
Consider the following reaction where Kp = 10.5 at 350 K.
2CH2Cl2(g) CH4(g) + CCl4(g)
If the...

Chemistry, 18.08.2020 20:01 alexcuevaz90
Consider the following reaction where Kp = 10.5 at 350 K.
2CH2Cl2(g) CH4(g) + CCl4(g)
If the three gases are mixed in a rigid container at 350 K so that the partial pressure of each gas is initially one atm, what will happen?
Indicate True (T) or False (F) for each of the following:
A. A reaction will occur in which CH2Cl2(g) is consumed.
B. Kp will decrease.
C. A reaction will occur in which CH4 is produced.
D. Qp is less than Kp.
E. The reaction is at equilibrium.
F. No further reaction will occur.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
The concept of empiricism states that all rationally accepted knowledge is determined from experience. francis bacon was one of the first scientists to promote this theory. what was it’s impact on society?
Answers: 1


Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2

Chemistry, 22.06.2019 15:00
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1
You know the right answer?
Questions

English, 25.09.2021 17:30

Social Studies, 25.09.2021 17:30


Mathematics, 25.09.2021 17:30


Chemistry, 25.09.2021 17:40

English, 25.09.2021 17:40


Advanced Placement (AP), 25.09.2021 17:40

Mathematics, 25.09.2021 17:40

Mathematics, 25.09.2021 17:40

English, 25.09.2021 17:40


Biology, 25.09.2021 17:40

Mathematics, 25.09.2021 17:40


Mathematics, 25.09.2021 17:40



English, 25.09.2021 17:40