

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 06:30
Design techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 2


Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1
You know the right answer?
What is the limiting reactant for the reaction below given that you start with 10.0 grams of Al (mol...
Questions


Social Studies, 20.01.2021 03:20

Mathematics, 20.01.2021 03:20

Mathematics, 20.01.2021 03:20

Mathematics, 20.01.2021 03:20

Arts, 20.01.2021 03:20

Computers and Technology, 20.01.2021 03:20

Mathematics, 20.01.2021 03:20


Mathematics, 20.01.2021 03:20

Mathematics, 20.01.2021 03:20

English, 20.01.2021 03:20

Mathematics, 20.01.2021 03:20


Mathematics, 20.01.2021 03:20


English, 20.01.2021 03:20

Mathematics, 20.01.2021 03:20


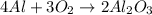
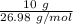
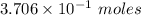
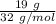
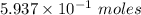
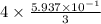

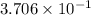 moles of Al available.
moles of Al available.

