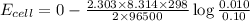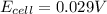Chemistry, 13.08.2020 18:01 scottcounts757
A concentration cell is one in which both the anode and cathode are the same but with different concentrations. Calculate the cell potential with [Zn2+] = 0.10 M[Zn2+] = 0.10 M for the cathode and the [Zn2+] = 0.010 M[Zn2+] = 0.010 M for the anode?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1
You know the right answer?
A concentration cell is one in which both the anode and cathode are the same but with different conc...
Questions


Mathematics, 09.06.2021 20:50







Mathematics, 09.06.2021 20:50

Mathematics, 09.06.2021 20:50



Chemistry, 09.06.2021 20:50

World Languages, 09.06.2021 20:50

Mathematics, 09.06.2021 20:50


Social Studies, 09.06.2021 20:50


English, 09.06.2021 20:50

Mathematics, 09.06.2021 20:50

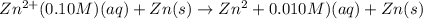
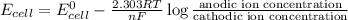
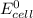 = standard electrode potential of the cell = 0 (as both metals are same )
= standard electrode potential of the cell = 0 (as both metals are same )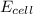 = emf of the cell = ?
= emf of the cell = ?