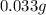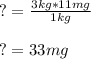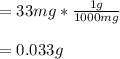Chemistry, 13.08.2020 04:01 adellbakarralus232
11mg of cyanide per kilogram of body weight is lethal for 50% of domestic chickens. If a chicken weighs 3kg, how many grams of cyanide would it need to ingest to kill 50% of domestic chickens?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the number of moles of chemical units represented by 9.03x10^24? and how do i show work? (dumb it down )
Answers: 1

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3
You know the right answer?
11mg of cyanide per kilogram of body weight is lethal for 50% of domestic chickens. If a chicken wei...
Questions






English, 26.01.2021 18:40


Mathematics, 26.01.2021 18:40

Mathematics, 26.01.2021 18:40




Mathematics, 26.01.2021 18:40




Social Studies, 26.01.2021 18:40



Mathematics, 26.01.2021 18:40