Answers: 1


Another question on Chemistry


Chemistry, 21.06.2019 20:00
2h2s + 3o2 2so2 + 2h2o which option gives the correct mole ratios? h2s: so2 = 2: 2 and o2: h2o = 3: 2 h2s: so2 = 2: 3 and o2: h2o = 3: 2 h2s: so2 = 4: 4 and o2: h2o = 5: 4 h2s: so2 = 4: 6 and o2: h2o = 4: 4
Answers: 1

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1
You know the right answer?
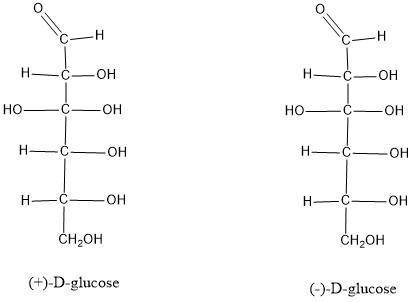
Why are(±)-glucose and (-)-glucose both classified as D sugar...
Questions

Health, 11.11.2020 01:30


Social Studies, 11.11.2020 01:30

Mathematics, 11.11.2020 01:30


Mathematics, 11.11.2020 01:30

Business, 11.11.2020 01:30

Chemistry, 11.11.2020 01:30

Mathematics, 11.11.2020 01:30

English, 11.11.2020 01:30





Mathematics, 11.11.2020 01:30


Arts, 11.11.2020 01:30

Mathematics, 11.11.2020 01:30