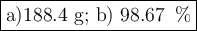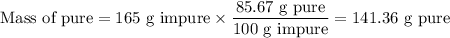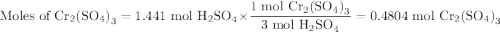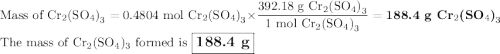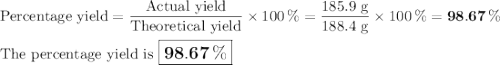Chromium is dissolved in sulfuric acid according to the following equation: Cr + H2SO4 ⇒ Cr2 (SO4) 3 + H2
a) How many grams of Cr2 (SO4) 3 can be obtained by reacting 165 g of 85.67% H2SO4 of purity?
b) If 485.9 g of Cr2 (SO4) 3 are obtained, what is the yield of the reaction?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Order the following from smallest to largest atom, electron, quark, proton, neutron, molecule, nucleus
Answers: 1

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3
You know the right answer?
Chromium is dissolved in sulfuric acid according to the following equation: Cr + H2SO4 ⇒ Cr2 (SO4) 3...
Questions

Mathematics, 08.09.2019 12:10





Law, 08.09.2019 12:10

Social Studies, 08.09.2019 12:10

Mathematics, 08.09.2019 12:10



Mathematics, 08.09.2019 12:10


Mathematics, 08.09.2019 12:10




History, 08.09.2019 12:10



Biology, 08.09.2019 14:10