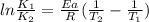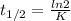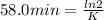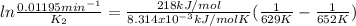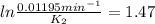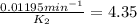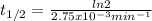Chemistry, 12.08.2020 05:01 alexmoy45p8yd7v
The decomposition of ethylene oxide(CH₂)₂O(g) → CH₄(g) + CO(g)is a first order reaction with a half-life of 58.0 min at 652 K. The activation energy of the reaction is 218 kJ/mol. Calculate the half-life at 629 K.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Consider the following system at equilibrium. caco3(s) ca2+(aq) + co32–(aq) the addition of which compound will cause a shift in equilibrium because of a common ion effect? ccl4 co2 cuso4 na2co3
Answers: 3

Chemistry, 22.06.2019 05:00
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3

Chemistry, 23.06.2019 06:00
Give one example of a pure (exact) number and of an estimated (measured) number.
Answers: 2
You know the right answer?
The decomposition of ethylene oxide(CH₂)₂O(g) → CH₄(g) + CO(g)is a first order reaction with a half-...
Questions



Mathematics, 05.02.2021 21:50





Arts, 05.02.2021 21:50

Mathematics, 05.02.2021 21:50


Arts, 05.02.2021 21:50


Mathematics, 05.02.2021 21:50

Mathematics, 05.02.2021 21:50

Mathematics, 05.02.2021 21:50

Physics, 05.02.2021 21:50


Mathematics, 05.02.2021 21:50

Mathematics, 05.02.2021 21:50

Mathematics, 05.02.2021 21:50