
Chemistry, 12.08.2020 05:01 hunterallan569
What is the frequency of a photon having an energy of 4.91 × 10–17 ? (c = 3.00 × 108 m/s, h = 6.63 × 10–34 J · s)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3

Chemistry, 21.06.2019 19:30
How many molecules of sucrose c12h22o11 are there in 454 grams of sucrose
Answers: 1

Chemistry, 21.06.2019 20:50
Choose all that apply. when creating a graph, you should: determine the x- and y- variables label the scale on the x- and y- axes plot the data points draw a line of best fit to represent the data trend
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1
You know the right answer?
What is the frequency of a photon having an energy of 4.91 × 10–17 ? (c = 3.00 × 108 m/s, h = 6.63 ×...
Questions


Biology, 04.07.2019 05:30

Biology, 04.07.2019 05:30


Mathematics, 04.07.2019 05:30







Health, 04.07.2019 05:30

English, 04.07.2019 05:30



History, 04.07.2019 05:30



Mathematics, 04.07.2019 05:30

History, 04.07.2019 05:30

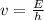
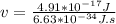
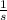 = 7.41*10¹⁶ Hz
= 7.41*10¹⁶ Hz

