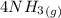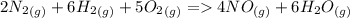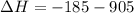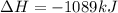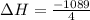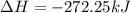Chemistry, 12.08.2020 06:01 Jazminfun70
Nitric oxide (NO) can be formed from nitrogen, hydrogen and oxygen in two steps. In the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) In the second step, ammonia and oxygen react to form nitric oxide and water: (g) (g) (g) (g) Calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. Round your answer to the nearest .

Answers: 1


Another question on Chemistry




Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3
You know the right answer?
Nitric oxide (NO) can be formed from nitrogen, hydrogen and oxygen in two steps. In the first step,...
Questions


Mathematics, 31.07.2020 04:01

Advanced Placement (AP), 31.07.2020 04:01


Mathematics, 31.07.2020 04:01






Mathematics, 31.07.2020 04:01





History, 31.07.2020 04:01

English, 31.07.2020 04:01



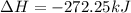 for 1 mole of NO.
for 1 mole of NO.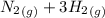 =>
=> 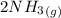
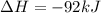
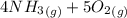 =>
=> 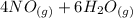

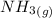 ) appeares as product in the first equation and as reagent in the 2 reaction, so when adding both, there is no need to inverse reactions. However, in the 2nd, there are 4 moles of that molecule, so to cancel it, you have to multiply by 2 the first chemical equation and enthalpy:
) appeares as product in the first equation and as reagent in the 2 reaction, so when adding both, there is no need to inverse reactions. However, in the 2nd, there are 4 moles of that molecule, so to cancel it, you have to multiply by 2 the first chemical equation and enthalpy: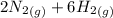 =>
=>