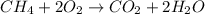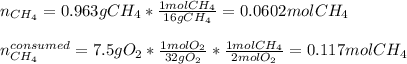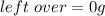Gaseous methane (CH4) will react with gaseous oxygen (O2) to produce gaseous carbon dioxide (CO) and gaseous water (H2O) . Suppose 0.963 g of methane is mixed with 7.5 g of oxygen. Calculate the minimum mass of methane that could be left over by the chemical reaction. Be sure your answer has the correct number of significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 13:30
A48 g piece of ice at 0.0 ∘c is added to a sample of water at 7.4 ∘c. all of the ice melts and the temperature of the water decreases to 0.0 ∘c. how many grams of water were in the sample?
Answers: 1

Chemistry, 21.06.2019 17:10
The concept of empiricism states that all rationally accepted knowledge is determined from experience. francis bacon was one of the first scientists to promote this theory. what was it’s impact on society?
Answers: 1

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 02:30
Based on the equation and the information in the table, what is the enthalpy of the reaction?
Answers: 2
You know the right answer?
Gaseous methane (CH4) will react with gaseous oxygen (O2) to produce gaseous carbon dioxide (CO) and...
Questions

Spanish, 12.03.2021 04:00


Mathematics, 12.03.2021 04:00




Mathematics, 12.03.2021 04:00

English, 12.03.2021 04:00

Mathematics, 12.03.2021 04:00




Mathematics, 12.03.2021 04:00



Mathematics, 12.03.2021 04:00


Advanced Placement (AP), 12.03.2021 04:00

Social Studies, 12.03.2021 04:00

World Languages, 12.03.2021 04:00